

Lactobacillus acidophilus Rosell-52, recently reclassified as Lactobacillus helveticus Rosell-52, is a very well-known strain of acidophilus. The strain is registered in the prestigious 'Collection National de Culture de Microorganisms (CNCM)' at the Institut Pasteur in France, and was named by leading probiotic laboratories, Institut Rosell, which manufactures the strain and has subjected it to extensive testing. Lactobacillus acidophilus Rosell-52 is very often researched along with Lactobacillus rhamnosus Rosell-11, and in combination these strains are often known by the name ‘Lacidofil®'.
Lactobacillus acidophilus Rosell-52 is a gram positive, homofermentative, facultative anaerobe. This is typically a rod shaped, non-sporulating strain that tends to form pairs or short chains. (Cocconcelli, 1997). As a β-galactosidase positive strain, it acts like lactase in the digestive system, and therefore may help to digest lactose.
The strain has been extensively used in many areas of research and found to have a wide range of potential benefits for gastrointestinal health, including its anti-pathogenic effects against undesirable bacteria such as Helicobacter pylori and Clostridium difficile. In combination with Lactobacillus rhamnosus Rosell-11, it has been shown to be one of the best strains to use for Antibiotic-Associated Diarrhoea (AAD), with this probiotic combination also being shown to withstand the damaging effects of the antibiotic treatment. Lactobacillus acidophilus Rosell-52 has also been featured in studies to explore the effects of probiotics in areas of health outside the digestive system, including immune function and mental health issues. It’s also interesting to note that a number of clinical trials using this probiotic strain have been conducted in young children, demonstrating its safety at various stages of life.

Lactobacillus acidophilus Rosell-52 is found in food supplements, often in combination with other strains. Its ability to survive gastric challenges such as stomach acid and biliary salts have been tested in vivo by microbiology Institute Rosell Lallemand. Such tests have demonstrated that L. acidophilus Rosell-52 is resistant to gastric acidity, with survival rates being optimal at pH 3 and above. They have also shown that Lactobacillus acidophilus Rosell-52 is able to survive high concentrations of bile, reporting that this strain should reach the gut alive.
A gold standard trial by Nagulesapillai, V. et al, (2015), used quantitative PCR methods to detect and quantify specific strains in faecal samples taken from participants who had taken a combination of two strains, including L. acidophilus Rosell-52 alongside antibiotic treatment. Tests showed L. acidophilus Rosell-52 was detected in 80% of faecal samples, showing excellent gastric survival rates.
The safety of this food supplement has also been investigated. Graheg-Ogden, D. (2018) conducted a risk assessment of Lactobacillus acidophilus Rosell-52 as an ‘other substance’ according to the food supplement directive 2002/46/EC. The report was written based on previous risk assessments and articles on the strain, concluding ‘it is unlikely that L. helveticus Rosell-52 ND causes adverse health effects in the general healthy population with a mature gastro-intestinal tract’.
In 2017 Manzano, S. et al, conducted a multi-centre, randomised, double-blind, placebo-controlled trial looking at the safety and tolerance of 3 strains in healthy infants, including L. acidophilus Rosell-52. The study on 221 healthy, full term infants aged 3-12 months reported no impact on growth, no serious adverse effects were experienced and any minor adverse events which occurred were equal across all groups, meaning that all studied strains, including L. acidophilus Rosell-52 are safe and well tolerated by infants.
This supports a study by Stojkovic, A. et al, (2016), which also reported good tolerance in young children under 5 years of age.
The Stojkovic, A. et al (2016) study trialled a food supplement containing 3 strains, including L. acidophilus Rosell-52, on children under 5 years, which resulted in an improvement to the children’s wellbeing, particularly in relation to respiratory comfort.
Safety studies conducted in infants & children are particularly valuable as they present the safety and tolerability in what is considered a more vulnerable population, going further to support the strains safety.
Antibiotics are one of the most widely-used drugs in medicine today, often with life-saving effects. For them to be fully effective, it is necessary for patients to complete the course of medication; however, antibiotics may prompt a variety of unpleasant side effects which can be so debilitating that many patients are unable to complete the treatment. Uncompleted courses of antibiotics are believed to be a cause of ‘antibiotic resistance’, as if not fully eradicated, pathogenic bacteria may mutate and become resistant to the drugs. Finding a remedy to alleviate side effects and enable patients to complete their courses of medication is therefore very important, and so probiotics have been explored as one possible solution.
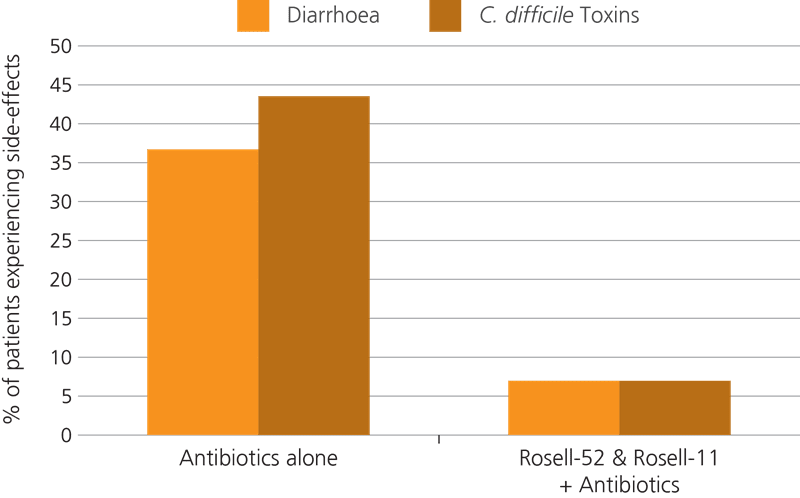
In a gold standard, randomised double-blind, placebo-controlled clinical trial using 146 healthy adults, Lactobacillus acidophilus Rosell-52 was tested alongside the Lactobacillus rhamnosus Rosell-11 strain to assess their combined potential against antibiotic-associated diarrhoea. All subjects were given a broad spectrum antibiotic for one week, and a treatment group was also given a probiotic supplement to take alongside the drugs, and then for a further seven days after antibiotic treatment had ceased. The control group were given a placebo. The results showed that the probiotic significantly shortened the duration of AAD symptoms (Evans et al 2016).
A second trial was conducted using 244 children aged 0-17 years, who were all suffering from either acute respiratory, urinary, or digestive infections requiring antibiotic treatment. The subjects were split into two groups, with 117 subjects being given antibiotics in combination with a supplement containing Lactobacillus acidophilus Rosell-52 and Lactobacillus rhamnosus Rosell-11, and the other 127 subjects being given antibiotic therapy alone. Again, the results showed a lower incidence and decreased duration of AAD in the probiotic group, with an additional benefit of decreased Clostridium difficile levels noted (Maydannik V.et al., 2010).
Numerous other multicentre, randomised, placebo-controlled, double-blind trials have tested the efficacy of this probiotic combination (Lactobacillus acidophilus Rosell-52 and Lactobacillus rhamnosus Rosell-11) for the prevention of AAD in adults. In a trial using 214 subjects who were already receiving antibiotic treatment for respiratory tract infections, the participants were randomised to receive either the probiotic or a placebo for 14 days. All subjects recorded bowel frequency and stool consistency daily. Results illustrated that members of the probiotic group had reduced bowel frequency compared to those in the placebo group (Song et al., 2010).
An earlier trial conducted in 2005 used 57 children who were taking antibiotics. The children were split into two groups, with one group being given the Lacidofil® supplement. The results indicated that 36.5% of those in the control group went on to develop antibiotic-associated diarrhoea, whereas only 7.4% of those in the probiotic group suffered this side effect (Ivanko O. & Radutnaya E., 2005).
Further relevant studies: Aryayev M. & Kononenko N. (2009),Dial S. et al., (2003), Doder R. et al., (2013), D’Souza A. L. et al., (2002), Gnaytenko O. et al., (2009), Jirapinyo, P. et al., (2002), Liskovich, V. et al., (2010), Marushko Y. & Shef G., (2007), Maydannik V. et al., (2008), Patsera M. et al., (2010).
Irritable Bowel Syndrome (IBS) is an umbrella term for a variety of different digestive symptoms, including bloating, cramps, diarrhoea, and constipation. The underlying cause of the condition is not clear, and the term is usually applied generically to these symptoms by medical professionals once any serious medical causes have been ruled out. At this point, doctors may prescribe various medications to help manage the symptoms. Research is linking the condition with the health of the gut flora, however, and probiotic supplements are becoming an increasingly popular natural adjunct or alternative to conventional interventions, and most of the featured strains in this database have been researched for this purpose. As part of the Lacidofil® supplement, Lactobacillus acidophilus Rosell-52 has been tested to try and determine whether it may offer any benefits in this condition.
A pilot study did indicate that this probiotic supplement may help to alleviate the symptoms of IBS. The trial used 50 patients who had been diagnosed with, and had suffered from, chronic IBS for at least ten years. The patients continued with their conventional medication but added in a daily probiotic supplement containing Lactobacillus acidophilus Rosell-52 and Lactobacillus rhamnosus Rosell-11. They were then monitored for a variety of digestive outcomes including changes in regularity, stool consistency and abdominal pain and distension. After four months of using the probiotic, 88% of the patients showed an overall reduction in IBS-related symptoms (Benes, Z. et al., 2006).
A more recent prospective, double-blind, randomised, controlled clinical trial was conducted using 76 adult patients. The subjects had all been suffering from acute diarrhoea, which in most cases was non-infectious. The patients were divided into two groups: a treatment group in which members were given Lacidofil for seven days, and a control group given a placebo supplement. The study found that the probiotic supplement shortened the duration of diarrhoea, and a statistically significant improvement in stool consistency was recorded. A small improvement in symptoms of abdominal pain, nausea, vomiting, bloating, headache, fever and tenesmus) was also noted (Simadibrata et al., 2013).
Further relevant studies: Chayka V. et al., (2006), Firmesse, O. et al., (2005), Kocian, J. et al., 1994, Rampengan et al., 2010, Skorodumova et al., (2007), Ten Bruggencate, (2015), Tlaskal, P. et al., (1995), Tlaskalet al., (2005), Wasowska-Krolikoeska K. et al., (1997), Zvyagintzeva et al., (2008).
Due to the increasing prevalence of mental health issues in the world’s population, extensive research is being conducted to try and determine the underlying physiological causes. A role for the gut flora and probiotics has been suggested, prompting a growing interest in the ‘gut-brain’ connection. In this area of probiotic research, Lactobacillus acidophilus Rosell-52 has also been tested in combination with Bifidobacterium longum Rosell-175, in supplement known sometimes as Probio’ Stick.
In June 2017 a small pilot study of 10 participants, with diagnosed depressive disorders, aimed to evaluate the effects of Lactobacillus acidophilus Rosell-52 and Bifidobacterium longum Rosell-175 on depression symptoms, such as low mood, anhedonia (inability to feel pleasure) and sleep disturbance. Participants were assessed using the Montgomery-Asberg depression rating scale to monitor mood symtoms, and sleep was monitored by observing brain activity, heart rate, and eye movement during the night. Participants supplemented with probiotics showed significant improvement in their symptoms, including improved sleep quality, mood score, and the ability to feel pleasure (Wallace C, et al., 2017).
A randomised, double blind, placebo-controlled trial took 55 healthy participants and gave them a supplement containing either the above probiotic combination, or a placebo, for a period of thirty days. The results were measured using various validated scales of anxiety and stress as well as depression and ‘coping’ scales. The findings showed that the use of this combined probiotic significantly reduced the scores of anxiety, depression and stress, and could have beneficial psychological effects in humans. A pre-clinical study in which the probiotic supplement was administered to rats also indicated that the Lactobacillus acidophilus Rosell-52 and Bifidobacterium longum Rosell-175 combination had anxiolytic-like activity (anti-anxiety effect) (Messaoudi et al., 2011).
An earlier double-blind, placebo-controlled, randomised study was conducted on participants with symptoms of stress. The subjects received either the Probio’ Stick probiotic combination (37 participants) or a placebo (38 participants) for three weeks. It was found the probiotic supplement significantly reduced stress-induced gastrointestinal symptoms, such as abdominal pain and nausea/vomiting, in the treatment group (Diop et al., 2008).
A multicentre, randomized, double-blind, placebo-controlled trial published in 2024 investigated the effects of L. helveticus Rosell-52 in combination with B. longum Rosell-175 on depressive symptoms in 116 adults diagnosed with major depressive disorder (Wallace et al., 2024). The intervention was administered for 60 days and outcomes were assessed using the Montgomery–Åsberg Depression Rating Scale (MADRS), Generalized Anxiety Disorder 7-item scale (GAD-7), and Perceived Stress Scale (PSS). While mean group differences between probiotic and placebo arms did not reach statistical significance, a greater proportion of subjects in the probiotic group achieved a clinically relevant reduction in MADRS scores.
Further relevant study: Yamamura S, et al. (2009)
Helicobacter pylori (H. pylori) is a species of bacteria that is often found residing in the human body, and it is thought to be present in around two thirds of the world's population. It typically lives in the digestive tract and, in most people, does not cause any noticeable symptoms; however, it can be a common cause of stomach ulcers.
It can be difficult to eradicate H. pylori once it becomes an issue, as it burrows into the stomach wall and 'hides'. The standard medical treatment from the doctor is a 'triple therapy' of two different antibiotics in combination with an antacid known as a proton pump inhibitor (PPI), but the treatment is not always completely successful. It is thought that probiotics may firstly help to reduce any negative side effects of the antibiotics, but may also have anti-pathogenic effects that could aid in eradicating this undesirable bacteria and make the treatment more effective.
A study was designed to evaluate the efficacy of the standard triple anti-H-pylori therapy both with, and without, a probiotic combination taken alongside the medication. To test this, 152 H. pylori-positive symptomatic patients were given the Lacidofil supplement alongside their 10 day triple therapy, and results were compared to a control group who had received the conventional treatment without a probiotic. Tests were conducted 6 weeks after therapy and it was found that those in the control group experienced a 72% success rate for eradication of the bacteria; however, in those subjects who had received the triple therapy plus the probiotic, the H. pylori eradication rate increased up to 92% (Bielanski et al., 2002).
A randomized, double-blind, placebo-controlled trial conducted in 2023 evaluated the effect of co-administering L. helveticus Rosell-52 and L. rhamnosus Rosell-11 with standard triple therapy for Helicobacter pylori eradication in symptomatic adults (Kiattiweerasak et al., 2023). The probiotic group demonstrated a statistically significant increase in eradication rates compared with the control group receiving antibiotics alone. In addition, participants receiving the probiotic combination experienced a reduction in the frequency and severity of gastrointestinal side effects typically associated with antibiotic therapy. These data extend previous findings on AAD by confirming the protective role of these strains during antibiotic exposure and indicate that they may also potentiate the therapeutic efficacy of eradication regimens.
Further relevant studies: Abaturov et al., (2014), Babak O., (2007), Gnaytenko et al., (2009), Plewinska E., (2006), Vdovychenko V., (2008), Ziemniak, W., (2006).
As approximately 70% of the cells involved in our immune responses are located in our intestinal wall, the interaction between our resident microbiota and the immune system has been the subject of considerable scientific interest. Research has indicated that there is a significant interplay between the gut flora and the immune system, and a potential role for probiotics in regulating and enhancing immune function. Lactobacillus acidophilus Rosell-52 is one of the probiotic strains that has been featured in associated studies.
In a randomised, double-blind, placebo-controlled study which was performed on 135 children aged 3-7 years during the winter months, Lactobacillus acidophilus Rosell-52 was used as part of a probiotic supplement that also included the strains Bifidobacterium infantis Rosell-33, Bifidobacterium bifidum Rosell-71 and a prebiotic in the form of Fructooligosaccharides (FOS). It was found that this probiotic combination specifically reduced the incidence of gastric disorders, and ear-nose-throat (ENT) or bronchopulmonary infections by 25%, with an associated 40% reduction in number of days absent from school due to sickness (Cazzola, et al. 2010).
Further relevant studies: Cazzola, M. et al., (2010), Marschan E. et al (2008)
Lactose intolerance is an increasingly common digestive issue, caused when the body is unable to digest the milk sugar lactose. As probiotic bacteria may help to break down lactose in the digestive system, their use to help alleviate this condition is being explored. The Lacidofil supplement is one of the probiotic combinations being researched for this purpose.
A small study using 19 patients with lactose intolerance attempted to demonstrate the efficacy of this probiotic supplement when given alongside dairy products such as milk, cottage cheese, and Edam cheese. Lactose tolerance and stool consistency were recorded both before and after the supplementation period of two weeks. The results indicated that Lactobacillus acidophilus Rosell-52 combined with Lactobacillus rhamnosus Rosell-11 helped to improve tolerance to the dairy foods by one third; this improvement in lactose tolerance was particularly significant when consuming milk (Kocian J., 1994).
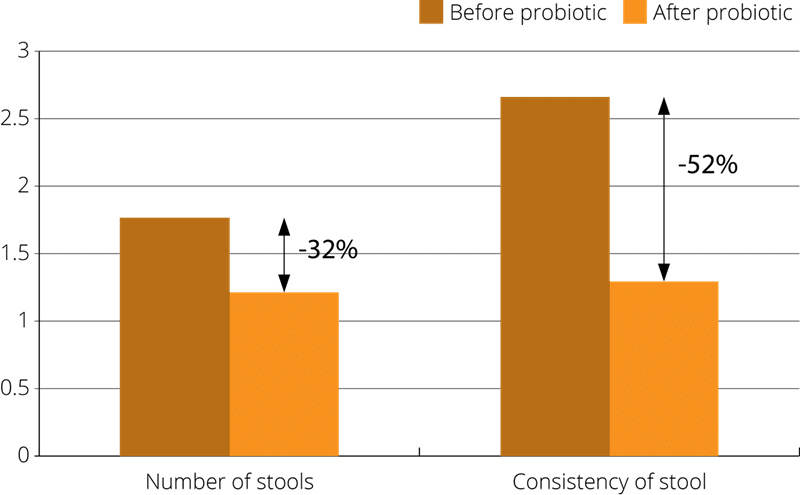
Further relevant studies: Chernyshov, (2007), Chernyshov, (2009a). Chernyshov, (2009b) Kocian J., (1996).
Vaginal dysbiosis (an imbalance of good & bad bacteria), typically presenting in the form of vaginal thrush, may be another unpleasant side effect of antibiotic treatment. Building on the outcome of previous research for antibiotic-associated diarrhoea, a 2010 study explored whether taking a probiotic supplement may also help to prevent vaginal symptoms in women taking antibiotics. A total of 96 women were used in the study; they had all undergone Caesarean section and were being given preventative antibiotics for seven days following the surgery. There were 56 women in the treatment group, all of whom were given a probiotic containing Lactobacillus acidophilus Rosell-52 and Lactobacillus rhamnosus Rosell-11 alongside their antibiotics, and a control group of 40 women who were given antibiotics only.
After the treatment, the integrity of the vaginal flora was seen to be preserved in 89.3% of the probiotic group, and no incidence of AAD was recorded. By comparison, in the control group a disruption in the vaginal flora was noted in all cases, and there was a 10% incidence of diarrhoea (Liskovich et al., 2010).
Further relevant studies: Chayka et al., (2006).
Authors: Information on this strain was gathered by Joanna Scott-Lutyens BA (hons), DipION, Nutritional Therapist; and Kerry Beeson, BSc (Nut.Med) Nutritional Therapist.
Last updated - 21st May 2020
As some properties & benefits of probiotics may be strain-specific, this database provides even more detailed information at strain level. Read more about the strains that we have included from this genus below.
Lactobacillus acidophilus strains: Lactobacillus acidophilus LA-05, Lactobacillus acidophilus NCFM®.
Lactobacillus casei strains: Lactobacillus casei Shirota, Lactobacillus casei DN-114001.
Lactobacillus plantarum strains: Lactobacillus plantarum LP299v.
Lactobacillus reuteri strains: Lactobacillus reuteri Protectis and Lactobacillus reuteri RC-14®.
Lactobacillus rhamnosus strains: Lactobacillus rhamnosus LGG®, Lactobacillus rhamnosus HN001, Lactobacillus rhamnosus GR-1® and Lactobacillus rhamnosus Rosell-11.
Lactobacillus paracasei strains: Lactobacillus paracasei CASEI 431®.
For more information and the latest research on probiotics, please visit the Probiotic Professionals pages.
For products containing this strain visit the Optibac Probiotics shop
Abaturov et al., (2014), ‘Efficiency of probiotic therapy in children with antibiotic-associated bowel disorders and Helicobacter pylori infection’. Sovremennaya pediatriya, 2:58.
Aryayev M. & Kononenko N., (2009), ‘Prevention of antibiotic-associated diarrhoea in patients with cystic fibrosis’. Odessa Medical Journal, 114(4):78.
Babak O. (2007), ‘The use of Lacidofil in treatment of duodenal peptic ulcers associated with H. pylori’. News of Pharmacy and Medicine, 5:24-25.
Benes Z. et al., (2006), ‘Lacidofil (Lb. acidophilus Rosell-52 and Lb. rhamnosus Rosell-11)
alleviates the symptoms of IBS’. Nutrafoods, 5:20-27.
Bielanski W. et al., (2002), ‘Improvement of anti-Helicobacter pylori therapy by the use of commercially available probiotics’. Gut, 5(11):A98.
Cazzola M. et al., (2010), ‘Immunomodulatory impact of synbiotic in TH1 and TH2 models of infection’. Therapeutic Advances in Respiratory Disease, 0(0):1-13.
Cazzola M. et al., (2010) ‘Efficacy of a synbiotic supplementation in the prevention of common diseases in children: a randomized, double-blind, placebo-controlled pilot study’ Therapeutic Advances in Respiratory Disease, 0(0):1-8.
Chayka V. et al., (2006), ‘Prevention of disbacteriosis in pregnant and women recently confined with surgical delivery’. News of Medicine and Pharmacy, 19:14-15.
Chernyshov, (2007), ‘Integrated treatment of infants, patients with atopic dermatitis. Dermatology, 3:23-26.
Chernyshov, (2009a), ‘B7-2/CD28 costimulatory pathway in children with atopic dermatitis and its connection with immunoglobulin E, intracellular interleukin-4 and in****eron-gamma production by T cells during a 1 month follow-up’. Journal of the European Academy of Dermatology and Venereology, 23:656-659.
Chernyshov, (2009b). ‘Randomized, placebo-controlled trial on clinical and immunologic
effects of probiotic Lacidofil® in infants with atopic dermatitis’. Microbial Ecology in Health and Disease, 21:228-232.
Cocconcelli, P.S et al., (1997), ‘Use of RAPD and 16S rDNA sequencing for the study of Lactobacillus population dynamics in natural whey culture’. Letters in Applied Microbiology, 25:8–12.
Dial S. et al. (2003), ‘Evaluation of the effectiveness of the probiotic Lacidofil (‘For those on antibiotics’) in the prevention of antibiotic associated diarrhoea: pilot study’. Montreal, Canada.
Diop L. & Guillou S., (2008), ‘Probiotic food supplement reduces stress-induced gastrointestinal symptoms in volunteers: a double-blind, placebo-controlled, randomized trial’. Durand Nutrition Research, 28(1):1-5.
Doder R. et al., (2013), ‘Outcomes of Clostridium difficile enterocolitis after administration of antibiotics along with probiotic supplement’. Medicinski Pregled., 66(5-6):209-13.
D’Souza A. L. et al., (2002), ‘Probiotics in prevention of antibiotic associated diarrhoea: meta-analysis’. British Medical Journal, 324:1362-1368.
Evans M. et al., (2016), ‘Effectiveness of Lactobacillus helveticus and Lactobacillus rhamnosus for the management of antibiotic-associated diarrhoea in healthy adults: a randomised, double-blind, placebo-controlled trial’. British Journal of Nutrition, 16(1):94-103.
Firmesse O. et al., (2005), ‘Quantification after transit in human digestive tract of ‘For those on antibiotics’ (Lactobacillus Rhamnosus Rosell 11 & Lactobacillus Acidophilus Rosell 52) consumed in a food supplement’. National Institute of Agronomic Research, Presented at Conference on Probiotics, Rome.
Firmesse O. et al., (2008), ‘Lactobacillus rhamnosus R11 consumed in a food supplement survived human digestive transit without modifying microbiota equilibrium as assessed by real-time polymerase chain reaction’. J Mol Microbiol Biotechnol., 14(1-3):90-9.
Foster T.A. et al., (2011), ‘A comprehensive post-market review of studies on a probiotic product containing Lactobacillus helveticus R0052 and Lactobacillus rhamnosus R0011’. Beneficial Microbes, 2(4):319-334.
Gnaytenko O. et al., (2009), ‘Antibiotic-associated diarrhoea as a complication of antihelicobacter therapy in children’. Practical Medicine, 5:76-83.
Ivanko O. & Radutnaya E., (2005), ‘Lactobacillus acidophilus reduces frequency of diarrhoea caused by toxins Clostridium difficile A+B in children treated by antibiotics’. Zaporozhye Medical Periodical, 2:21-23.
Jirapinyo P. et al., (2002), ‘Prevention of antibiotic-associated diarrhoea in infants by probiotics’. J Med Assoc Thai.; 85 (2):S739-S42.
Kiattiweerasak R., Sirimontaporn A., Wisedopas N. et al., (2023), ‘Adjunctive probiotic therapy with Lactobacillus helveticus Rosell-52 and Lactobacillus rhamnosus Rosell-11 improves eradication rates in Helicobacter pylori-positive patients receiving standard triple therapy: a randomized controlled trial’. Helicobacter, 28(5):e12955.
Kocian J. & Lekarstvi V., (1994), ‘Lactobacilli in treatment of dyspepsia in dysmicrobia of different etiology’, Vnitrni Lekarstvi, 40(c2):S79-83.
Kocian J., (1996), ‘Further Possibilities in the Treatment of Lactose Intolerance: Lactobacilli’ Department of Internal Medicine Institute for Post-gradual Education of Medical Staff’, Prague, Czech Republic, Jun 14, 2003.
Lallemand Health Solutions internal data, 2012
Liskovich, V. et al.(2010), ‘Efficiency of Lacidofil-WM for prevention of vaginal dysbiosis and antibiotics-associated diarrhoea in puerperas after caesarean operation’. Health, 1:63-66.
Manzano S et al., (2017) Safety and tolerance of three probiotic strains in healthy infants: a multi-centre randomized, double-blind, placebo-controlled trial. Beneficial Microbes 1-10 online
Marschan E. et al., (2008), ‘Probiotics in infancy induce protective immune profiles that are characteristic for chronic low-grade inflammation’. Clinical and Experimental Allergy, 38: 611–618.
Marushko Y. & Shef G., (2007), ‘Current status of antibiotics-associated bowel disorders issue in children’. Perinatology & Paediatrics, 4:65-68.
Maydannik V. et al., (2008), ‘Prevention and treatment of antibiotic-associated diarrhoea’. Paediatrics Obstetrics & Gynaecology, 1:63-65.
Maydannik V. et al., (2010), ‘Efficiency and safety of Lacidofil in children with antibiotic-associated diarrhoea caused by Clostridium difficile’. Pediatrics, Obstetrics & Gynaecology, 3:53-57.
Messaoudi M. et al., (2011), ‘Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects’. British Journal of Nutrition, 105(5):755.
Nagulesapillai, V. et al (2015) Detection and quantification of strain specific probiotics in clinical faecal samples of healthy adults on antibiotic treatment by quantitative PCR.
Patsera M. et al., (2010), ‘Using probiotic strains Lactobacillus acidophilus R0052 and Lactobacillus rhamnosus R0011 during pulmonary tuberculosis in children, complicated by antibiotic-associated Clostridium difficile intestinal infection’. Zaporozhye Medical Journal, 12:30-33.
Plewinska E. et al., (2006), ‘Probiotics in the treatment of Helicobacter pylori infection in children’. Gastroenterolgia Polska, 13(4):315-319.
Rampengan et al., (2010), ‘Comparison of efficacies between live and killed probiotics in children with lactose malabsorption’. Southeast Asian Journal of Tropical Medicine and Public Health, 41:474-481.
Simadibrata M, Ndraha S, Tedjasaputra R, Syam AF, Santi AFA, Rani A. Revealing the effect of probiotic combination Lactobacillus rhamnosus and Lactobacillus acidophilus (Lacidofil®) on acute diarrhea in adult patients. Journal of Clinical Medicine and Research 2013;5(2):23-8.
Song H. et al., (2010), ‘Effect of probiotic Lactobacillus (Lacidofil® Cap) for the prevention of antibiotic-associated diarrhoea: a prospective, randomised double-blind multicenter study’. Journal of Korean Medical Science, 25:1784-1791.
Stojkovic, A. et al (2016) Clinical trial / experimental study (consort compliant): Optimal time period to achieve the effects on synbiotic-controlled wheezing and respiratory infections in young children. Serbian journal of management. 144 (1-2):38-45
Wallace C. et al., (2017), Findings presented at 13th World Congress of Biological Psychiatry. Poster P-05-015, presented June 19, 2017.
Wallace C. et al., (2024), ‘Probiotic supplementation with Lactobacillus helveticus Rosell-52 and Bifidobacterium longum Rosell-175 in patients with major depressive disorder: results from the PRO-DEMET randomized controlled trial’. Journal of Psychiatric Research, 174:120–129.