

Lactobacillus casei is one of numerous species belonging to the Lactobacillus genus. As of April 2020 L. casei has been officially reclassified to Lacticaseibacillus casei so the full strain name may also be referred to as Lacticaseibacillus casei Shirota (Zheng J et al., 2020). Strains from this particular species are often present in fermented foods such as sauerkraut and yoghurt, and the Lactobacillus casei Shirota strain is typically found in commercially available yoghurt drinks.
The strain has been widely researched to explore its potential to offer support for a wide variety of health and digestive conditions, including immune function, infectious diarrhoea, and functional intestinal disorders such as Irritable Bowel Syndrome (IBS).
In particular, Lactobacillus casei Shirota has been investigated in relation to easing diarrhoea caused by unknown gut infections, known infections such as Clostridium difficile and Helicobacter pylori, viruses such as norovirus and rotavirus and antibiotics.
Lactobacillus casei Shirota containing fermented milk product has been consumed for several decades. The ability of L. casei Shirota to survive the gut has been tested in numerous human studies. A study demonstrated that consumption of L. casei Shirota-fermented milk (100 ml containing 1 billion cfu, three times daily for 4 weeks) resulted in an increase of the Lactobacillus count (10 million cfu/g) in the faeces suggesting that this strain can survive through the gut environment (Spanhaak, S. et al., 1998). However, after the ingestion period, the levels of L. casei Shirota decreased and reverted back to their pre-intervention state within 2 weeks. In another study, the survival of L. casei Shirota was tested using both culture and a strain specific monoclonal antibody to identify the cultured colonies (ELISA) (Tilley, L. et al., 2014). The number of viable Lactobacillus casei Shirota in the faeces was shown to increase when consuming the fermented milk drink containing L. casei Shirota and decreased during wash-out. Mai, T. et al., (2017) demonstrated the gastrointestinal survival of L. casei Shirota in 26 healthy Vietnamese adults when a fermented milk drink containing 100 million cfu/ml of the Lactobacillus casei strain was consumed daily for 14 days. After 1 and 2 weeks of ingestion, L. casei Shirota was recovered from all 26 faecal samples at an average of 50 million and 54 million CFU/g faeces, respectively. Another study conducted in an Asian population showed that the levels of L. casei Shirota at approx. 6.6 ± 0.6 log10 CFU/g were detected in faeces in all volunteers after consumption of fermented milk containing L. casei Shirota (1 billion cfu daily) for ten days (Utami, T. et al., 2015).
Lactobacillus casei Shirota is taken as a food by consumers worldwide daily, and with this comes a certain level of assumed safety. We can also look to clinical documentation for an assessment of its safety. One trial looked at the safety of this strain in a particularly vulnerable group of children in the paediatric intensive care unit in the UK (Srinivasan, R. et al., 2006). Twenty-eight subjects were randomised to receive L. casei Shirota in a dose of 10 million cfu/day in three divided doses via a nasogastric tube for a maximum duration of 5 days. Safety was assessed by looking for evidence of translocation of this strain in cultures obtained from study subjects, of which none were found. In addition there were no apparent side effects or adverse events reported. We can see from this study in a vulnerable group that the use of L. casei Shirota appears safe and well tolerated, and therefore in otherwise healthy people we can be confident of a good safety profile.

Antibiotics are widely used medications but often have unpleasant side effects, such as antibiotic-associated diarrhoea (AAD). Their use is also associated with an increased risk of Clostridium difficile infection (CDI). It is believed that the infections occur due to the negative effects of antibiotics on the delicate balance of the intestinal flora. Lactobacillus casei Shirota has been used in various studies to assess its potential to preserve the balance of flora and possibly prevent diarrhoea symptoms.
With this focus, a large trial observed 678 hospital patients who were taking a broad range of different antibiotics. For the purposes of the study, the subjects were divided into two groups: those in the treatment group were given Lactobacillus casei Shirota in a fermented drink, and those in the control group given a placebo, both during their antibiotic treatment and for three days afterwards. The results showed that intervention with Lactobacillus casei Shirota reduced the antibiotic-induced decrease in abundance of total gut bacteria and Bifidobacterium observed in the placebo group. Lactobacillus casei Shirota intervention also increased levels of Lactobacillus bacteria (Pirker et al., 2012).
A further single site, cohort-control study of 66 patients with Clostridium difficile infection (CDI) attempted to determine whether Lactobacillus casei Shirota could help to prevent or alleviate Clostridium difficile infection (CDI). The subjects were all being treated with either antibiotics alone or antibiotics and probiotics (L. casei Shirota). It was noted that the recurrence of CDI was much lower in the probiotic group suggesting that the strain L. casei Shirota can help reduce the recurrence of Clostridium difficile infection (Lee L.Y.W., 2013).
A Japanese study, performed using 77 elderly patients in a residential home setting, attempted to determine whether a probiotic supplement could reduce the risk of viral gastroenteritis outbreaks. During the study, half of the participants were given a fermented drink containing Lactobacillus casei Shirota, and the other half were given a placebo drink, and the supplementation took place over the winter months. The results indicated that those taking the probiotic had reduced fever symptoms compared to those taking a placebo. Whilst the incidence of norovirus was unchanged, stool tests revealed a significant increase in the presence of Bifidobacterium and Lactobacillus after one month on the probiotic. Notably, after two months of supplementation, there was a decrease in levels of the largely pathogenic bacterial groups Enterobacteriaceae and Pseudomonas (Nagata et al., 2011).
Lactobacillus casei Shirota was also trialled in a group of elderly subjects with norovirus gastroenteritis in a nursing home setting, to ascertain if the use of a probiotic could help to normalise bowel movements and improve infection control. This was a placebo controlled trial with 35 participants in each group. A significant difference in the intestinal microbiota, including a reduction in the numbers of destructive bacteria such a Clostridium difficile, an improvement in bowel movements and a reduction in fever was seen in the probiotic group compared to the placebo group (Nagata S, et al 2016).
It is becoming well documented that a healthy balance of gut bacteria is important for the prevention and alleviation of constipation. Research has indicated that both the types and levels of intestinal flora in constipated people differ to those typically found in healthy people (Quigley E.M., 2011). The ability of beneficial bacteria to produce short chain fatty acids and metabolise bile salts is also very significant in maintaining regular and efficient stool transit.
To test the effects of probiotic supplementation on constipation symptoms, a double-blind, placebo-controlled, randomised trial used 120 adult patients from Belgian doctor’s surgeries, who had all been diagnosed with mild constipation. There was a four week intervention period during which a treatment group was supplemented with a fermented milk drink containing Lactobacillus casei Shirota, while a control group was given a placebo drink. The results illustrated that, compared to the placebo group, the participants taking the L. casei Shirota probiotic drink experienced a significant improvement in stool consistency, with their stools becoming softer and easier to pass. Viable Lactobacillus casei Shirota was also found in their stool tests (Tilley et al., 2014).
The L. casei Shirota strain was also trialled in a double-blind, placebo-controlled, randomised study which was conducted over a four-week period, using 70 chronically constipated adults. All of the subjects had experienced symptoms of constipation, flatulence and bloating. The subjects were randomised to receive either a placebo, or a probiotic drink containing Lactobacillus casei Shirota. The results indicated that 89% of the probiotic group experienced an improvement in constipation symptoms and stool consistency, compared to 56% of the placebo group; however, no change was observed in flatulence or bloating symptoms (Koebnick et al., 2003).
Other Relevant Studies: Cassani E. et al., (2011), Krammer H.J. et al., (2011), Matsumoto K. et al., (2010), Matsumoto K. et al., (2006), Sakai T. et al., (2011), Sakai T. et al., (2015), Shioiri T. et al., (2006), Tilley L. et al., (2014), Van den Nieuwboer M. et al., (2015).
Irritable Bowel Syndrome (IBS) is an umbrella term used to diagnose those suffering from a myriad of gastrointestinal symptoms. These include abdominal pain, bloating, diarrhoea, constipation, and flatulence, though symptoms may vary. The aetiology of IBS is still relatively unknown; however, microbial imbalance and small intestinal bacterial overgrowth (SIBO) are believed to be factors in the development of IBS symptoms. Consequently, the use of probiotics as a useful intervention is being explored.
A randomised, double blind, placebo controlled trial involving 80 patients with IBS looked at the effects of probiotic supplementation on their symptoms. A probiotic drink containing Lactobacillus casei Shirota was administered to subjects over an eight week period. The results showed a 30% improvement on clinically relevant IBS symptoms in those taking the probiotic (Thijssen A.Y. et al., 2011).
Other Relevant Studies: Barrett I.S. et al., (2008).
Diarrhoea symptoms are particularly concerning when they present in young children, as they may become easily dehydrated by persistent loose stools. Easily applied interventions to prevent diarrhoea are therefore of particular interest to scientists, and probiotics have been explored as a possible solution.
A very large double-blind, placebo-controlled, randomised study involving 3758 children living in a slum community in India, attempted to evaluate this potential. Every day for 12 weeks, the child volunteers were given either a probiotic drink containing Lactobacillus casei Shirota (LcS), or a placebo drink. After assessing the results, the study authors concluded that probiotics appeared to reduce the risk of diarrhoea by 14%, and that this strain of bacteria could help to significantly reduce the incidence of symptoms in children living in a high-risk environment (Sur D. et al., 2010).
There are believed to be several ways in which probiotics may help modulate the immune system, and as you will see from those featured in this database, different strains may have an effect on different aspects of immune function. Lactobacillus casei Shirota has been shown to increase the activity of natural killer (NK) cells, which are large lymphocytes that are part of the innate immune system. They circulate in the blood targeting abnormal cells such as certain tumour cells and viral-infected cells.

This beneficial effect was demonstrated in a double-blind, placebo-controlled, randomised study involving 72 Italian working class smokers. During the three week test period, half of the subjects were given a probiotic supplement containing Lactobacillus casei Shirota, and the other half were given a placebo. Those taking the probiotic had a significantly increased NK activity, which would otherwise have been decreased due to cigarette smoking (Reale M. et al., 2011).
Salivary immunoglobulin A (s-IgA) is a mucosal antibody that plays an important role in protecting our bodies from upper respiratory tract infections (URTI). Reduced production of this antibody appears to be associated with physical or mental stress, and low s-IgA levels are found in those who suffer from recurrent infections. Studies conducted with Lactobacillus casei Shirota in athletes have shown that consumption of this probiotic can help maintain salivary IgA levels, and thus reduce incidence of colds.
In a double blind, placebo-controlled trial conducted during the winter months, 84 highly active individuals were randomised to receive either a probiotic containing Lactobacillus casei Shirota or a placebo. Results indicated that s-IgA concentration was elevated in the probiotic group compared to placebo (and baseline results). The authors concluded that regular ingestion of Lactobacillus casei Shirota appears to reduce the frequency of URTI in athletes, which may be related to better maintenance of salivary IgA levels (Gleeson M. et al., 2011).
Other Relevant Studies: Cats A. et al., (2003), Chiba Y. et al., (2010), Dong et al., (2013), Eguchi S. et al., (2010), Falasca K. et al., (2015), Fujita R. et al., (2013), Gleeson M. et al., (2011), Kanazawa H. et al., (2005), Man E.R. et al., Matsuzaki T., et al., (2005), Morimoto K. (2005), Motoori M. et al., (2015), (2013), Nagao F. (2000), Nagata S. et al., (2016), Sgouras D. et al., Shida K., (2011), Shida K. et al., (2006a), Shida K. et al., (2006b), Stadbauer V. et al., (2008), Sugawara G. et al., (2006), (2004), Takeda K., (2007), Thomson C.H. et al., (2012), Wong S. et al., (2015), Wong S. (2014), Yasuda E., (2008).
The onset of allergy symptoms are thought to be an indication of an unbalanced immune system. An allergic response results from an imbalance of Th1:Th2 cells, causing an overreaction to harmless substances resulting in symptoms such as rhinitis, eczema, asthma etc. It has been found that infants with allergies often have an imbalanced gut microbiota with higher numbers of Clostridia, fewer Bifidobacteria and with species more typical of those in adults.
A randomised double-blind, placebo-controlled study attempted to investigate the effects of Lactobacillus casei Shirota in patients with allergic rhinitis triggered by Japanese cedar pollen (JCP). For a period of eight weeks, subjects in the treatment group were given fermented milk containing L. casei Shirota whilst those in the control group were given a placebo. Clinical symptoms and immunological parameters were monitored and compared between the two groups, and it was noted that, whilst the probiotic supplements did not prevent the allergy symptoms, they appeared to reduce nasal symptoms (Tamura et al., 2007)
A double-blind, placebo-controlled trial investigated a possible role for Lactobacillus casei Shirota (LcS) in the modulation of seasonal allergic rhinitis (SAR). This small study observed 20 volunteers who were split into treatment and control groups. Over a period of five months, the subjects in the treatment group were given a milk drink fermented with Lactobacillus casei Shirota, whereas the control group were given a plain milk placebo drink. Changes in immune status were monitored and documented in each group, and the results indicated that those in the probiotic group displayed a significant reduction in levels of antigen-induced IL-5, IL-6 and IFN-gamma production compared with volunteers supplemented with placebo. Additionally, levels of specific IgG increased and IgE decreased in the treatment group, and the study authors concluded that the probiotic helped to modulate immune responses in allergic rhinitis, and could help to reduce the severity of symptoms (Ivory et al., 2008).
Other Relevant Studies: Akoglu et al., (2015), Almeida C. et al., (2012), Bian L. (2011), Braga T. et al., (2011), Candy D. et al., (2001), Endo H, et al., (2011), Fujimori S. et al., (2007), Mitsuyama K. et al., (2008), Shimizu M. et al., (2012), Tsuji H. et al., (2014), Wong S. et al., (2014), Wong S. et al., (2015).
Stress is an increasingly common issue in the modern world, with doctors being inundated with requests for solutions to help manage the often very debilitating symptoms of anxiety and depression. Due to medication side effects, options for the best natural adjuncts are being explored, and due to the increasingly well-documented link between gut health and mental health, there is much interest in a role for probiotics in the management of mental health.
In a double-blind, placebo-controlled trial, 50 medical students were tested to demonstrate the effects of the probiotic Lactobacillus casei Shirota (LcS) strain on the psychological, and physical stress responses triggered when undertaking an examination. In association with an increase in anxiety experienced one day before the examination, salivary cortisol and plasma L-tryptophan levels were found to be significantly increased only in the placebo group. Additionally, two weeks after the examination, the Lactobacillus casei Shirota group had significantly higher faecal serotonin levels than the placebo group, suggesting a higher production of this neurotransmitter (low serotonin levels are associated with anxiety). It was also noted that incidence of cold and abdominal symptoms were significantly lower in the Lactobacillus casei Shirota group than in the placebo group during the pre-examination period. These results suggest that the daily consumption of fermented milk containing Lactobacillus casei Shirota may offer benefits for the prevention of stress-related physical symptoms in healthy subjects exposed to traumatic situations (Kato-Katoaka A. et al., 2016)
A further trial by the same authors was conducted on 23 subjects for 8 weeks before an examination to test if Lactobacillus casei Shirota could help to reduce stress-related digestive symptoms. A control group was made up of 24 students who were given a placebo. It was found that the probiotic helped to preserve the diversity of the gut bacteria and reduced abdominal symptoms of stress (Kato-Katoaka A. et al., 2016).
Although a common resident of the human body, Helicobacter pylori is considered to be a pathogen because, in some individuals, it can cause chronic inflammation of the stomach lining which leads to the development of stomach ulcers. The bacteria lies dormant in the stomach mucosal surface without causing symptoms unless circumstances allow it to proliferate and cause infection. It is one of the most common causes of stomach ulcers, and is usually treated with a triple therapy including two different types of antibiotics and a proton pump inhibitor. As this bacterial species can retreat into the stomach lining to escape the effects of the medication, the therapy is not always 100% effective, and probiotics have been considered as a natural adjunct to conventional medication in order to increase the success of the treatment.
A randomised comparative trial investigated whether there may be a benefit in adding a probiotic into the triple therapy protocol. Lactobacillus casei Shirota was the selected strain, and was administered alongside the conventional triple therapy in subjects receiving antibiotic treatment for H. pylori infection. Based on breath test measurements, eradication of H. pylori was achieved in 29 out of 31 of subjects given the probiotic (94%) compared to 25 out of 33 subjects in the control group (76%) (Sahagún-Flores J.E. et al., 2007).
Though the highest concentration of our resident bacteria are to be found in the intestines, the importance of a healthy and balanced flora in all areas of the body is now being highlighted. The composition of the oral flora and its implications for good oral health is becoming widely recognised, and over the past decade or so, a role for probiotics in the maintenance of oral healthcare has been considered.
A 2013 study assessed the benefits of this probiotic strain for oral health. Subjects were given a probiotic drink containing Lactobacillus casei Shirota for a period of four weeks. Whilst no significant changes to the populations of oral flora were noted, a temporary and intake-dependent presence of the L. casei Shirota LcS bacterial strain was observed. The study authors believed that this study might pave the way for future studies focusing on the use of probiotics to support subjects who were at greater risk of oral infection, as it was thought that the probiotic may reduce the levels of pathogens in the oral flora (Sutula J. et al., (2013).
Authors: Information on this strain was gathered by Joanna Scott-Lutyens BA (hons), DipION, Nutritional Therapist; and Kerry Beeson, BSc (Nut.Med) Nutritional Therapist.
Last updated: 25th May 2020
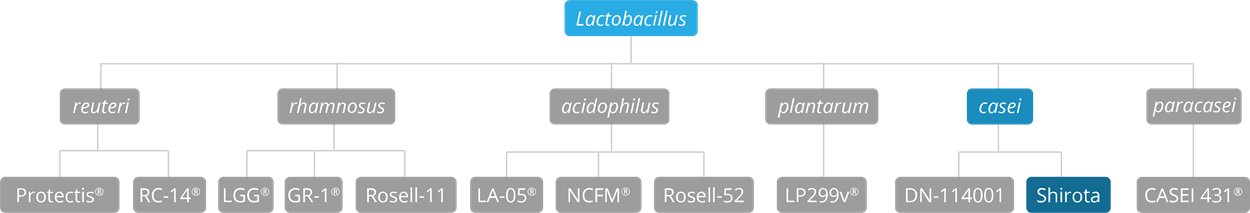
As some properties & benefits of probiotics may be strain-specific, this database provides even more detailed information at strain level. Read more about the strains that we have included from this genus below.
Lactobacillus acidophilus strains: Lactobacillus acidophilus LA-05, Lactobacillus acidophilus NCFM®, Lactobacillus acidophilus Rosell-52.
Lactobacillus casei strains: Lactobacillus casei DN-114001.
Lactobacillus plantarum strains: Lactobacillus plantarum LP299v.
Lactobacillus reuteri strains: Lactobacillus reuteri Protectis and Lactobacillus reuteri RC-14®.
Lactobacillus rhamnosus strains: Lactobacillus rhamnosus LGG®, Lactobacillus rhamnosus HN001, Lactobacillus rhamnosus GR-1® and Lactobacillus rhamnosus Rosell-11.
Lactobacillus paracasei strains: Lactobacillus paracasei CASEI 431®.
For more information and the latest research on probiotics, please visit the Probiotic Professionals pages.
For products containing these strains visit the Optibac Probiotics shop.
Akoglu B., et al., (2015), ‘Probiotic Lactobacillus casei Shirota improves kidney function, inflammation and bowel movements in hospitalized patients with acute gastroenteritis – A prospective study’. Journal of Functional Foods, 17:305-313.
Almeida C.C., et al., (2012), ‘Beneficial effects of long-term consumption of a probiotic combination of Lactobacillus casei Shirota and Bifidobacterium breve Yakult may persist after suspension of therapy in lactose intolerant patients’. Nutrition in Clinical Practice, 27(2):247-251.
Barrett J.S. et al., (2008), ‘Probiotic effects on intestinal fermentation patterns in patients with irritable bowel syndrome’. World Journal of Gastroenterology, 14(32):5020-5024.
Bian L. et al., (2011), ‘Effects of the continuous intake of Lactobacillus casei strain Shirota-fermented milk on risk management of long-term inpatients at health service facilities for the elderly’. International Journal of Probiotics and Prebiotics, 6(2):123-132.
Braga T.D. et al., (2011), ‘Efficacy of Bifidobacterium breve and Lactobacillus casei oral supplementation on necrotizing enterocolitis in very-low-birth-weight preterm infants: a double-blind, randomized, controlled trial’. American Journal of Clinical Nutrition, 93:81-86.
Candy D.C.A. et al., (2001), ‘Effect of administration of Lactobacillus casei Shirota on sodium balance in an infant with short bowel syndrome’. Journal of Pediatric Gastroenterology & Nutrition, 32(4):506-508.
Cassani E. et al., (2011), ‘Use of probiotics for the treatment of constipation in Parkinson’s disease patients’. Minerva Gastroenterologica e Dietologica, 57:117-121.
Chen L. et al. (2022), 'Probiotic supplementation attenuates age-related sarcopenia via the gut-muscle axis in SAMP8 mice'. Journal of Cachexia. Sarcopenia and Muscle, 13(1):515-531.
Chiba Y. et al., (2010), ‘Well-controlled proinflammatory cytokine responses of Peyer’s patch cells to probiotic Lactobacillus casei.’ Immunology, 130:352-362.
Dong H. et al., (2013), ‘Immunomodulatory effects of a probiotic drink containing Lactobacillus casei Shirota in healthy older volunteers’. European Journal of Nutrition, 52(8):1853-1863.
Eguchi S. et al., (2010), ‘Perioperative synbiotic treatment to prevent infectious complications in patients after elective living donor liver transplantation. A prospective randomised study’. American Journal of Surgery, 201(4):498-502.
Endo H. et al., (2011), ‘Efficacy of Lactobacillus casei treatment on small bowel injury in chronic low-dose aspirin users: a pilot randomized controlled study’. Journal of Gastroenterology, 46:894-905.
Falasca K. et al., (2015), ‘Effect of probiotic supplement on cytokine levels in HIV-infected individuals: a preliminary study’. Nutrients, 7:8335-8347.
Fujimori S. et al., (2007), ‘High dose probiotic and prebiotic co-therapy for remission induction of active Crohn’s disease’. Journal of Gastroenterology & Hepatology, 22: 1199- 1204.
Fujita R. et al., (2013), ‘Decreased duration of acute upper respiratory tract infections with daily intake of fermented milk: A multicentre, double-blinded, randomized comparative study in users of day care facilities for the elderly population’. American Journal of Infection Control, 41(12):1231-1235.
Gleeson M. et al., (2011), ‘Daily probiotic’s (Lactobacillus casei Shirota) reduction of infection incidence in athletes’. International Journal of Sport Nutrition & Exercise Metabolism, 21:55-64.
Ivory K. et al., (2008), ‘Oral delivery of Lactobacillus casei Shirota modifies allergen-induced immune responses in allergic rhinitis. Clinical and Experimental Allergy, 38:1-8.
Kato-Katoaka A. et al., (2016), ‘Fermented milk containing Lactobacillus casei strain Shirota prevents the onset of physical symptoms in medical students under academic examination stress’. Benefical Microbes, 7(2):153-156.
Kato-Katoaka A. et al., (2016), ‘Fermented milk containing Lactobacillus casei strain Shirota preserves the diversity of the gut microbiota and relieves abdominal dysfunction in healthy medical students exposed to academic stress’. Appl Environ Microbiol, 82(12):3649-58.
Kanazawa H. et al., (2005), ‘Synbiotics reduce postoperative infectious complications: a randomized controlled trial in biliary cancer patients undergoing hepatectomy’. Langenbeck's Arch Surgery 390(2): 104-113.
Koebnick C. et al., (2003), ‘Probiotic beverage containing Lactobacillus casei Shirota improves gastrointestinal symptoms in patients with chronic constipation’. Canadian Journal of Gastroenterology, 17(11):655 – 659.
Krammer H.J. et al., (2011), ‘Effect of Lactobacillus casei Shirota on colonic transit time in patients with chronic constipation’. Coloproctology, 33:109-113.
Lee L.Y.W. et al., (2013), ‘Prevention of relapse following Clostridium difficile infection using probiotic Lactobacillus casei Shirota’. International Journal of Probiotics & Prebiotics, 8(4):145- 148.
Mai, T. et al., (2017) ‘Recovery of Lactobacillus casei strain Shirota (LcS) from the intestine of healthy Vietnamese adults after intake of fermented milk’. Asia Pac J Clin Nutr, 26(1):72-77.
Mann E.R. et al., (2013), ‘Dysregulated circulating dendritic cell function in ulcerative colitis is partially restored by probiotic strain Lactobacillus casei Shirota’. Mediators of Inflammation, Article ID 573576.
Matsuzaki T. et al., (2005), ‘A prospective uncontrolled trial of fermented milk drink containing viable Lactobacillus casei strain Shirota in the treatment of HTLV-1 associated myelopathy / tropical spastic paraparesis’. Journal of the Neurological Sciences, 273:75-81.
Matsumoto K. et al., (2006), ‘The effect of a probiotic milk product containing Lactobacillus casei strain Shirota on the defecation frequency and the intestinal microflora of sub-optimal health state volunteers: a randomized placebo-controlled cross-over study’. Bioscience Microflora, 25(2):39-48.
Matsumoto K., (2010), ‘Effects of a probiotic fermented milk beverage containing Lactobacillus casei strain Shirota on defecation frequency, intestinal microbiota, and the intestinal environment of healthy individuals with soft stools’. Journal of Bioscience & Bioengineering, 110(5):547-52.
Mitsuyama K. et al., (2008), ‘Beneficial effects of Lactobacillus casei in ulcerative colitis: a pilot study’. Journal of Clinical Biochemistry and Nutrition, 43(S1):78- 81.
Motoori M. et al., (2017), ‘Randomized study of the effect of synbiotics during neoadjuvant chemotherapy on adverse events in esophageal cancer patients’. Clinical Nutrition, 36(1):93-99
Morimoto K. et al., (2005), ‘Modulation of natural killer cell activity by supplementation of fermented milk containing Lactobacillus casei in smokers’. Preventive Medicine, 40:589-594.
Nagao F., (2000), ‘Effects of a fermented milk drink containing Lactobacillus casei strain Shirota on the immune system in healthy subjects’. Bioscience Biotechnology & Biochemistry, 64(12):2706-2708.
Nagata S. et al., (2011), ‘Effect of the continuous intake of probiotic-fermented milk containing Lactobacillus casei strain Shirota on fever in a mass outbreak of norovirus gastroenteritis and the faecal microflora in a health service facility for the aged’. British Journal of Nutrition, 106(4):549-56.
Nagata S. et al., (2016), ‘The effectiveness of Lactobacillus beverage in controlling infections among the residents of an aged care facility: a randomized placebo-controlled double-blind trial’. Ann Nutr Metab, 68(1):51-59.
Nagata S. et al., (2011), ‘Effect of the continuous intake of probiotic-fermented milk containing Lactobacillus casei strain Shirota on fever in a mass outbreak of norovirus gastroenteritis and the faecal microflora in a health service facility for the aged’. Br J Nutr., 106(4):549-56.
Nagata S. et al., (2016), ‘The effectiveness of Lactobacillus beverage in controlling infections among the residents of an aged care facility: a randomized placebo-controlled double-blind trial’. Ann Nutr Metab., 68(1):51-59.
Pirker et al., (2012), ‘Effects of antibiotic therapy on the gastrointestinal microbiota and the influence of Lactobacillus casei’. Food & Agric Immunology, 24(3):315-330.
Quigley E.M., (2011), ‘The enteric microbiota in the pathogenesis and management of constipation’. Practical Res Clin Gastroenterol’., 25 (1):119-26.
Reale M, et al., (2011), ‘Daily Lactobacillus casei Shirota intake increases natural killer cell activity in smokers’. British Journal of Nutrition, 108(2):308-314.
Sakai T. et al., (2015), ‘Effect of fermented milk containing Lactobacillus casei strain Shirota on constipation-related symptoms and haemorrhoids in women during puerperium’. Beneficial Microbes, 6(3):253-262.
Sakai T. et al., (2011), ‘Fermented milk containing Lactobacillus casei strain Shirota reduces incidence of hard or lumpy stools in healthy population’. International Journal of Food Sciences & Nutrition, 62(4):23-30.
Sahagún-Flores J.E. et al., (2007), ‘Eradication of Helicobacter pylori: triple treatment scheme plus Lactobacillus vs triple treatment alone’. Cirug Ciruj 75(5): 333-336.
Sgouras D. et al., (2004), ‘In vitro and in vivo inhibition of Helicobacter pylori by Lactobacillus casei strain Shirota’. Applied & Environmental Microbiology, 70(1):518- 26.
Shida K. et al., (2017), ‘Daily fermented milk with Lactobacillus casei strain Shirota reduces the incidence and duration of upper respiratory tract infections in healthy middle-aged office workers’. Eur J Nutr, 56(1):45–53.
Shida K. et al., (2006), ‘Essential roles of monocytes in stimulating human peripheral blood mononuclear cells with Lactobacillus casei to produce cytokines and augment natural killer cell activity’. Clinical & Vaccine Immunology, 13(9):997-1003.
Shida K. et al., (2006), ‘Induction of interleukin-12 by Lactobacillus strains having a rigid cell wall resistant to intracellular digestion’. Journal of Dairy Science, 98:3306-3317.
Shida K., (2011), ‘Flexible cytokine production by macrophages and T cells in response to probiotic bacteria’. Gut Microbes, 2(2):109-114.
Shioiri T. et al., (2006), ‘The effects of a synbiotic fermented milk beverage containing Lactobacillus casei strain Shirota and transgalactosylated oligosaccharides on defecation frequency, intestinal microflora, organic acid concentrations, and putrefactive metabolites of sub-optimal health state volunteers: a randomized placebo-controlled cross-over study’. Bioscience Microflora, 25(4):137-146.
Shimizu M, et al., (2012), Successful treatment of primary sclerosing cholangitis with a steroid and a probiotic’. Case Reports in Gastroenterology, 6:249-253.
Spanhaak, S. et al., (1998) ‘The effect of consumption of milk fermented by Lactobacillus casei strain Shirota on the intestinal microflora and immune parameters in humans’. Eur J Clin Nutr, 52(12): 899-907.
Srinivasan, R. et al., (2006) ‘Clinical Safety of Lactobacillus casei shirota as a Probiotic in Critically Ill Children. Journal of Pediatric’. Gastroenterol Nutr, 42:171–173.
Stadlbauer V. et al., (2008), ‘Effect of probiotic treatment on deranged neutrophil function and cytokine responses in patients with compensated alcoholic cirrhosis’. Journal of Hepatology, 48:945-951.
Sugawara G. et al., (2006), ‘Perioperative synbiotic treatment to prevent postoperative infectious complications in biliary cancer surgery: A randomized controlled trial’. Annal of Surgery, 244(5):706-714.
Sur D. et al., (2010), Role of probiotic in preventing acute diarrhoea in children: a community-based, randomized, double-blind placebo-controlled field trial in an urban slum. Epidem Infect., 139(6):919-26.
Sur D. et al., (2010), ‘Role of probiotic in preventing acute diarrhoea in children: a community-based, randomized, double-blind placebo-controlled field trial in an urban slum’. Epidemiology and Infection, 139(6):919-926.
Sutula J., et al (2013), 'The effect of a commercial probiotic drink containing Lactobacillus casei strain Shirota on oral health in healthy dentate people', Microbiol. Ecol. Health Dis., 29:24 10.3402/mehd.v24i0.21003 [PMC free article] [PubMed].
Takeda K. & Okumura K., (2007), ‘Effects of a fermented milk drink containing Lactobacillus casei strain Shirota on the human NK-cell activity’. The Journal of Nutrition, 137(1): 791-793.
Takeda K. et al., (2006), ‘8 Interleukin-12 is involved in the enhancement of human natural killer cell activity by Lactobacillus casei Shirota’. Clinical & Experimental Immunology, 146:109-115.
Tamura M. et al., (2007), ‘Effects of probiotics on allergic rhinitis induced by Japanese cedar pollen: randomized double-blind, placebo-controlled clinical trial’ International Archives of Allergy & Immunology, 143(1):75-82.
Tilley et al., (2014), ‘A probiotic fermented milk drink containing Lactobacillus casei strain Shirota improves stool consistency of subjects with hard stools’. Int J Probiotics Prebiotics, 9(1/2):23-30.
Tilley L. et al., (2014), ‘A probiotic fermented milk drink containing Lactobacillus casei strain Shirota improves stool consistency of subjects with hard stools’. Int J Probiotics Prebiotics, 9(1/2):23-30.
Thijssen A.Y. et al., (2016), ‘Efficacy of Lactobacillus casei Shirota for patients with irritable bowel syndrome’. European Journal of Gastroenterology & Hepatology, 28(1):8-14.
Thijssen A.Y. et al., (2011), ‘A randomized, placebo-controlled double blind study to assess the efficacy of a probiotic dairy product containing Lactobacillus casei Shirota on symptoms of irritable bowel syndrome’. Gastroenterology, 140(5,S1):S609.
Thomson C.H. et al., (2012), ‘Yakult: a role in combating multi-drug resistant Pseudomonas aeruginosa?’ Journal of Wound Care, 21(11):566-569.
Tsuji H. et al., (2014), ‘Maintenance of healthy intestinal microbiota in women who regularly consume probiotics’. Int J Probiotics Prebiotics, 9(1/2):31-38.
Utami, T. et al., (2015) ‘Recovery of Lactobacillus casei strain shirota (LCS) from the intestine of healthy Indonesian volunteers after intake of fermented milk and its impact on the enterobacteriaceae faecal microbiota’. Inter J Probiotics Prebiotics, 10: 77-84.
Van den Nieuwboer M. et al., (2015), ‘Improving the bowel habits of elderly residents in a nursing home using probiotic fermented milk’. Beneficial Microbes, 6(4):397- 403.
Wong S. et al., (2015), ‘Use of probiotics in preventing antibiotic-associated diarrhoea and Clostridium difficile associated diarrhoea in spinal injury centres: an international multicentre study’. International Journal of Probiotics Prebiotics, 10(23):85-90.
Wong S.S. et al., (2014), ‘A Lactobacillus casei Shirota probiotic drink reduces antibiotic-associated diarrhoea in patients with spinal cord injuries: a randomized controlled trial’. British Journal of Nutrition, 111(4):672-678.
Yasuda E. (2008), ‘Suppressive effect on activation of macrophages by Lactobacillus casei strain Shirota genes determining the synthesis of cell wall-associated polysaccharides’. Applied & Environmental Microbiology, 74(15):4746-4755.
Zheng J, Wittouck S. et al., (2020) 'A taxonmonic note on the genus Lactobacillus: Description of 23 novel genera, emended description of the genus Lactobacillus Beijerinck 1901, and union of Lactobacillaceae and Leuconostocaceae'. Int.J.Syst.Evol.Microbiol, 70(4): 2782-2858. DOI: 10.1099/ijsem.0.004107