Etiquette often dictates that modesty is the best policy, however in certain situations, I think it is acceptable, or even advisable, to over-rule that and to sing your accomplishments from the proverbial rooftops! So, if you can excuse my immodesty, I’d like to take this occasion to do just that, and to ‘shout’ about the huge amount of scientific research that we have behind our live cultures supplements.
Best live cultures brand...?
As the following graph depicts, at Optibac we have significantly more clinical trials on our finished products, than any other live cultures brand available in the UK. This means that our customers can take our supplements with every confidence that they have been thoroughly researched! This is important as every bacterial strain has different characteristics and has different modes of action in our bodies. No two strains are equal, and for that reason we like to have concrete ‘evidence’ that our formulations really do lead the way in terms of research...
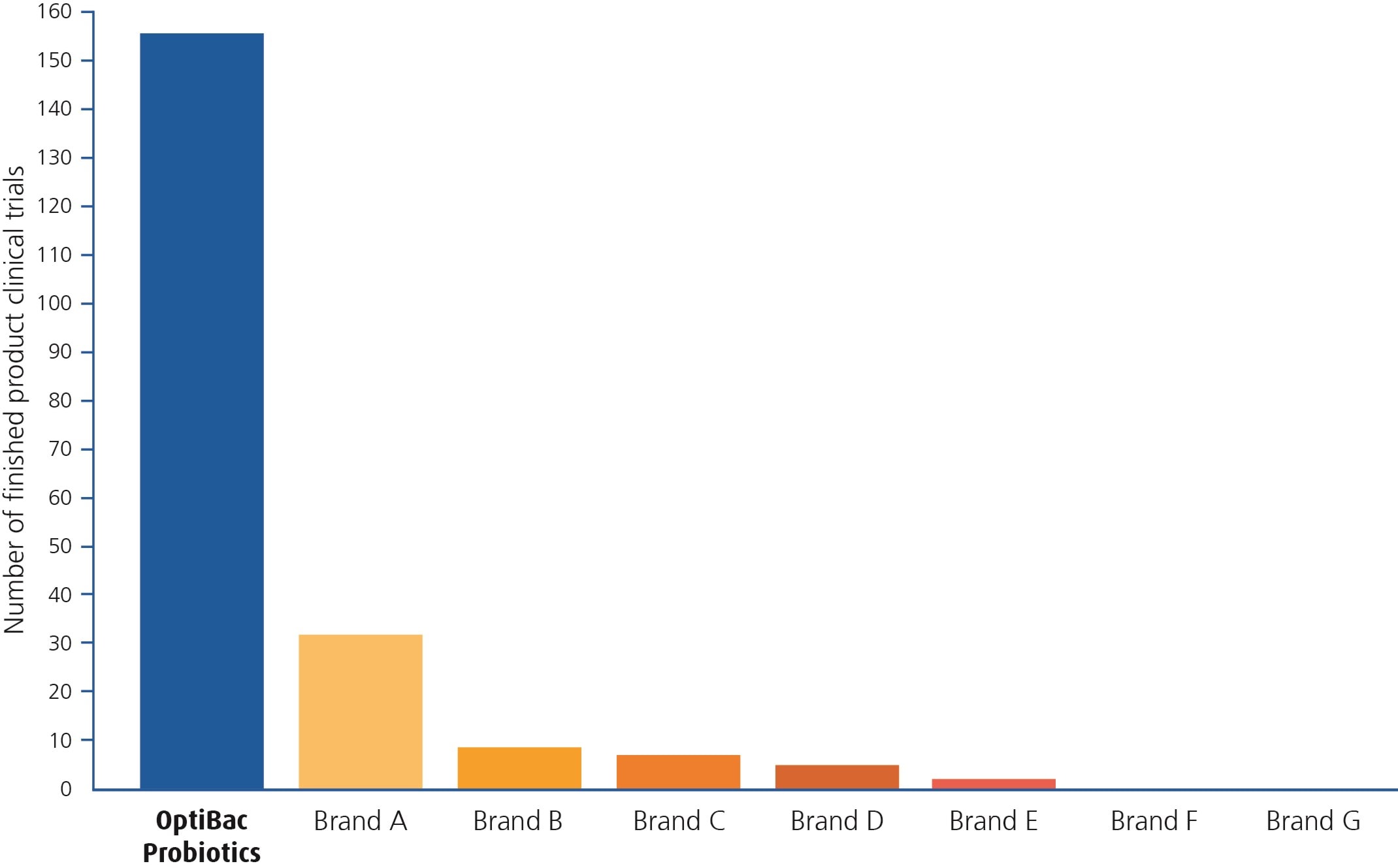
Optibac Probiotics versus other leading live cultures brands
I have omitted the brand names from the graph, as my aim is simply to demonstrate the extent to which we research our products before bringing them to market. It is not my intention to undermine other brands who may choose to have other priorities. So, suffice it to say then that brands A-G on the graph relate to the other leading live cultures brands in the UK. The number of clinical trials attributed to their ranges are genuine and accurate, and we spent a long time looking in depth at the research on everyone's products! The graph shows the numbers of trials that each brand has on finished products, so Optibac has 156 finished product clinical trials to date (a finished product trial is one conducted on the exact product; combination of strains and dose as sold in store). If you were to include all of the trials that we also have on single strains, or different combinations of strains within each product, then the number would be much higher.
Types of research
There are different types of research that a product can under-go, both in-vitro and in-vivo.
In-vitro tests are performed in a laboratory setting, using simulated conditions of the human GI tract. This type of testing should be performed to ensure that the live bacteria:
Survive at room temperature (even refrigerated live cultures spend time away from the fridge)
Resist gastric acidity & bile salts (so that they survive to reach the gut!)
Where applicable1, adhere to the intestinal wall (it is only then that friendly bacteria can multiply in the gut)
Inhibit pathogens once in the intestines.
All of the strains that we use have undergone this testing.
In-vivo tests on the other hand are carried out in living beings, and should ideally be performed in addition to the standard in-vitro tests that I have just mentioned. Human clinical trials are performed to understand if a certain friendly bacteria strain or combination of strains has a strong potential to confer health benefits to the host. A good live cultures supplement will have strain and formula-specific double-blind, randomised, placebo-controlled clinical trials that have been carried out in humans. As demonstrated above, the Optibac range has nearly five times more clinical trials than our nearest competitor. Furthermore, wherever possible our clinical trials are conducted on the group for whom we intend the product e.g. the trials on ‘For those on antibiotics’ have specifically tested this supplement in those taking antibiotics, and is likely the most researched live cultures supplement alongside antibiotics - tested alongside many different types of antibiotics and at various strengths.
Who funds our clinical trials?
Funding of trials can be an important question, as in the interest of impartiality, trials should ideally be performed by an independent third party with no vested interest in the clinical outcomes. The vast majority of trials on the Optibac finished products, and as on the blue bar in the graph above, are funded by universities and research institutions around the world, using resources from university funds, government funding and grants from organisations such as the EU.
Optibac: tried and tested
We are proud of that fact that our range of supplements has more finished product clinical trials than any other friendly bacteria brand in the UK. Our aim is to bring the best quality and most effective, strain-specific live cultures to the market, and clinical trials are an important component of this strategy. The information they provide allows us to target the strains we use, rather than adopting a 'one-formulation-fits-all' type of approach. We hope that this makes it easier for you to choose which product you or your clients would use, so that you can recommend them with confidence.
This blog was last updated 22/09/20.
Notes:
1. Adherence to the intestinal wall is not always applicable, depending on the live cultures strain in question. For example, the strain S. boulardii is understood to be transient, and therefore does not adhere and multiply in the gut, yet it still meets the essential criteria for a 'probiotic'; i.e. to have a positive benefit on the host. Furthermore, other strains such as L. reuteri RC-14® and L. rhamnosus GR-1®, as in Optibac 'For women' are actually tested to colonise the vaginal flora, and not the gut flora - therefore despite being some extremely well researched strains, these are not tested to adhere to the gut wall.