


Unique IS-2 is a microbial strain belonging to the Bacillus coagulans species (Please note, the strain was previously known as Lactobacillus sporogenes but has been reclassified to B. coagulans due to its spore forming abilities). B. coagulans Unique IS-2 is a gram positive, rod shaped and spore forming bacterium that can survive in both aerobic and anaerobic conditions (facultative anaerobe). This transient strain has the potential to produce a variety of digestive enzymes, anti-microbial molecules (including bacteriocins) and vitamins.
Researched in: children (aged 2+), adults and the elderly
B. coagulans Unique IS-2 has been shown in studies to be safe and reach the small intestine where it begins to germinate. All clinical trials outlined in this article found no significant differences between probiotic and placebo groups in terms of adverse events and safety.
A double blind, randomised, placebo controlled (gold standard) study found B. coagulans Unique IS-2 to be safe in 141 children with diagnosed IBS. No significant differences in adverse events were found between the probiotic and placebo group (Sudha et al., 2018). This was also confirmed in a study investigating 136 adults with IBS; no significant differences were noted between probiotic and placebo groups (Madempudi et al., 2019).
A highly advanced in vitro ‘Simulator of Human Intestinal Microbial Ecosystem (SHIME®)’ model was used to investigate survival rates and germination of B. coagulans Unique IS-2. The results showed excellent survival rates under both fasted and fed environments (99%+). B. coagulans Unique IS-2 began germination into the vegetative cell in the small intestine, and this continued into the colon. In the colon, the vegetative cells were able to produce lactic acid and acetate (also produced by lactic acid bacteria) (Ahire, Neelamraju and Madempudi, 2020).
The risk of children suffering from GI issues is increasing. A recent study in the US assessing 1,255 children found 24.7% of infants and toddlers and 25% of children and adolescents (aged 4-18) suffered with a functional GI disorder, which was also associated with a decreased quality of life (Robin et al., 2018). A child’s gut microbiome can differ from an adult’s, therefore positive probiotic trials in adults do not always correlate to children. B. coagulans Unique IS-2 has been shown to significantly improve a variety of GI disorders in children and adolescents.
A double blind randomised and placebo controlled (gold standard) study was conducted to assess the effects of B. coagulans Unique IS-2 in 141 children aged 4-12 and diagnosed with IBS according to Rome III criteria. The study was split into three phases. Phase one - a two-week run-in period, phase two - eight week treatment period and phase three - a two week wash out period. During phase two, children were divided into groups; group one received 2x109 of B. coagulans Unique IS-2 in a chewable format and group two received a placebo. They took their respective supplement daily. A range of IBS symptoms were assessed throughout the study period.
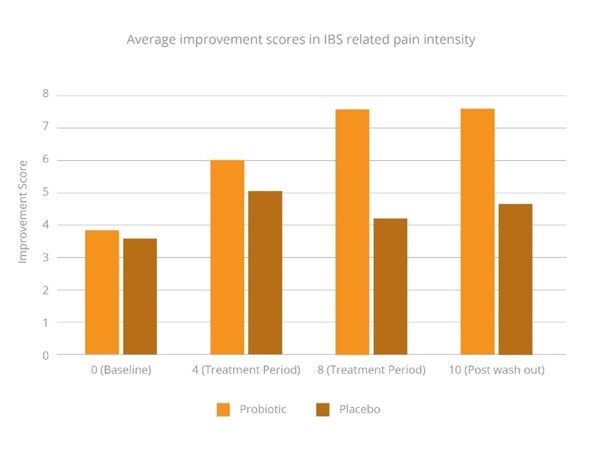
In the probiotic group, there was a significant improvement in pain intensity scores (Graph 1) compared to placebo. By week eight, 93% of participants in the probiotic group reported more than 50% relief in pain, compared with 22% in the placebo group.
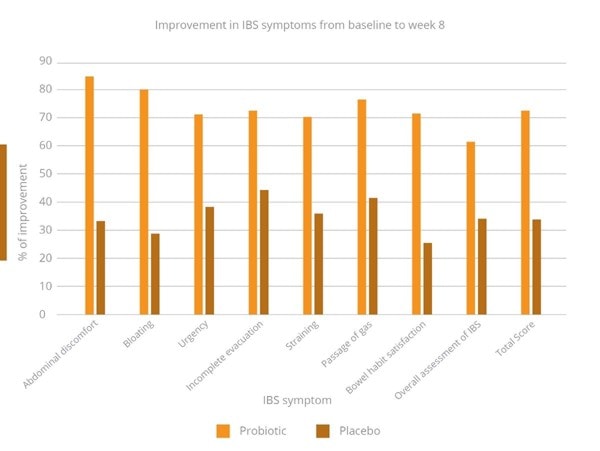
Significant improvements in a range of IBS symptoms (Graph 2) were seen in the probiotic group by week eight. These included; reduction in abdominal discomfort, bloating, straining, urgency, incomplete evacuation and passage of gas. Total scores also significantly reduced (data not shown). There was also a significant improvement in stool consistency by week eight in the probiotic group. Almost 50% of individuals in this group achieved normal stool consistency compared with 19% in the placebo group.
Results were also confirmed with further analysis using a globally accepted ‘Subject’s Global Assessment of Relief (SGARC)’ score. This data also showed significant improvements in ‘Family assessment of the impact of their child’s IBS on the family’s life’ in the probiotic group. All together these results highlight huge potential for B. coagulans Unique IS-2 on a child’s gut and digestive health and could benefit those with diagnosed IBS (Sudha et al., 2018).
A gold standard study investigated 115 children, diagnosed with FAP under the Rome III criteria. The children aged between 6-18 years were divided into two groups. Group one received a combination of B. coagulans Unique IS-2 (150 million CFU) and FOS (100mg), and Group two received a placebo, twice daily for four weeks. A follow up at 12 weeks was also included. Treatment response rates were analysed, response was defined as over a 2-point reduction in the 6-point self-rated pain scale or “no pain”.
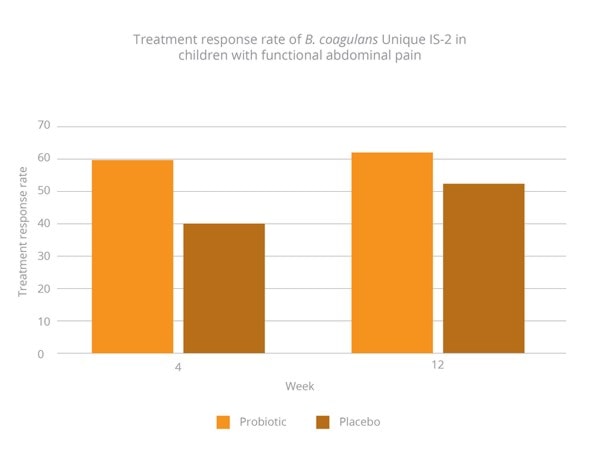
Response rate was significantly higher in the probiotic group compared with placebo at four weeks (60% v 40%) (Graph 3). At 12 weeks, results showed this trend had continued but was no longer significant (64.4% v 52.4%) (Graph 3). The physician-rated global severity score over the study period had also improved, almost reaching significance (p=0.06). Results highlight the potential for B. coagulans Unique IS-2 in children with FAP (Saneian et al., 2015).
A randomised controlled trial investigated the effects of B. coagulans Unique IS-2 in 120 children aged 4-13 years, diagnosed with abdominal pain using the Rome III criteria. Participants were divided into three groups. Group one received pH- dependent peppermint oil (Colpermin) three times daily, Group two received B. coagulans Unique IS-2 (150 million CFU) and FOS, three times a day and Group three took a placebo. The groups took their respective supplement for one month. Both interventions showed promising results. Pain duration and frequency significantly improved in the probiotic group compared to placebo (Asgarshirazi, Shariat and Dalili, 2015).
A controlled trial was conducted to assess B. coagulans Unique IS-2 in children with functional constipation diagnosed by Rome III criteria. The children aged between 2- 14 years were divided into two groups; Group one received 150 million CFU of B. coagulans Unique IS-2 per 20kg body weight per day plus mineral oil (1ml/kg/day). Group two received the mineral oil alone. Both groups took their respective supplement daily for eight weeks. Stool frequency, straining at defecation, incomplete evacuation and soiling (faecal incontinence) improved in both groups, however a greater improvement was noted in the probiotic group.
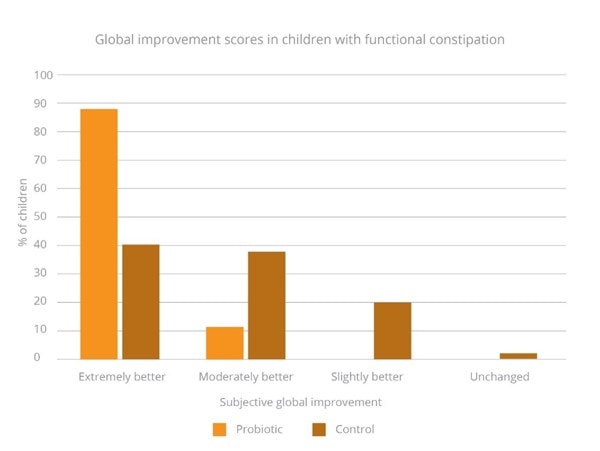
Results also showed a better global improvement in the probiotic group, 87% of individuals reported ‘Extremely better’ improvement compared with 40% in the control group (Graph 4). These results highlight the ability of B. coagulans Unique IS-2 to support individuals with constipation and IBS related constipation (Saneian et al., 2013).
It is estimated that on average 17% of adults in the UK suffer with IBS, with higher rates in women (23%) than men (11%) (Khanbhai and Singh Sura, 2013). IBS and gut health issues can impact quality of life and mental wellbeing. B. coagulans Unique IS-2 has been shown to improve IBS related symptoms and other GI symptoms.
A gold standard trial investigated B. coagulans Unique IS-2 in 136 adults who met the Rome III criteria for IBS. All participants underwent a two week wash out period and were then divided to receive either a placebo or B. coagulans Unique IS-2 (2 billion CFU) daily for eight weeks. Abdominal pain and bowel movements were primarily investigated, alongside other IBS symptoms and cytokine profiles.
B. coagulans Unique IS-2 was able to significantly reduce abdominal pain and increase the number of complete spontaneous bowel movements compared with placebo. At the end of week eight, in the probiotic group, pain scores halved from 8.2 to 3.4, compared with 8.3 to 6.7 in the placebo group. Additionally, 85% of individuals in the probiotic group reported more than 50% improvement in pain reduction compared with 13% in the placebo group. Bowel movement score increased from an average of 2.5 to 4 bowel movements per week in the probiotic group compared to placebo (2.2 to 2.4) by week eight.
From week five, those in the probiotic group exhibited significant improvements in abdominal discomfort, bloating, urgency, incomplete evacuation, straining, passage of gas, bowel habit satisfaction, overall assessment of IBS symptoms and total score. From week six, bowel consistency significantly improved in the probiotic group with 65% of individuals gaining normal consistency compared with 33% in the placebo group.
At the end of the study, it was found that almost 20% of individuals had complete and 64% had considerable relief from IBS symptoms in the probiotic group. This was significantly higher than placebo group (0% complete and 11% considerable) (Madempudi et al., 2019).
A gold standard study assessed B. coagulans Unique IS-2 in 100 adults with Rome III diagnosed functional constipation. Individuals were divided to receive 2 billion CFU of B. coagulans Unique IS-2 or a placebo daily for one month. At week three and four, significant improvements were found in bowel movements in the probiotic group compared with placebo. Movements had increased from an average of 0.9 in week one to 5.98 at week 6 compared with 0.94 to 3.12 in the placebo group. Stool consistency also significantly improved from week three in the probiotic group. Additionally, by the end of the study the feeling of incomplete evacuation significantly improved and a decrease in abdominal pain and defecation pain were noted in the probiotic group compared with placebo. The results highlight promise for B. coagulans Unique IS-2 to support those with constipation (Madempudi et al., 2020).
A small clinical trial was conducted in 28 adults with acute diarrhoea (three bowel movements within 24 hrs for less than seven days). All participants received 2 billion CFU of B. coagulans Unique IS-2 twice daily for 10 days. Duration of diarrhoea decreased from 35 mins to 3.5 mins per day and frequency of diarrhoea decreased from 7.96 to 0.76 times per day. Abdominal pain and consistency of stool also improved over the study. These results demonstrate the use of B. coagulans Unique IS-2 to alleviate diarrhoeal symptoms (Sudha and Bhonagiri, 2012).
Small intestinal bacterial overgrowth (SIBO) can cause complex GI symptoms. Although the condition and treatment are not well understood, probiotics have been considered to play an important role. A small gold standard trial investigated B. coagulans Unique IS-2 in 30 individuals with chronic abdominal pain or diarrhoea and a positive hydrogen breath test (HBT). All participants underwent three weeks of antibiotic therapy (using broad spectrum antibiotics). The individuals were then randomised to receive either B. coagulans Unique IS-2 or a placebo daily for 15 days alongside a maintenance antibiotic dose. After six months, HBT’s and GI symptoms were analysed.
In the probiotic group, abdominal pain had disappeared completely. Plus, GI symptoms including belching, flatulence and diarrhoea all significantly improved. HBT’s turned negative in 93% of individuals in the probiotic group compared with 67% in the placebo group. The study suggests potential for B. coagulans Unique IS-2 in those with SIBO, to reduce symptoms and enhance current treatments (Khanbhai and Singh Sura, 2013).
In vitro studies have highlighted a potential role for B. coagulans Unique IS-2 in immune system modulation and promoting anti-inflammatory effects (Sudha and Arunasree, 2015; Azimirad, Alebouyeh and Naji, 2017). A clinical trial was also conducted to assess the immune boosting effects of the probiotic in humans.
A gold standard study was conducted in 68 adults aged 58-67 years old. Participants were divided into three groups; Group 1 received a control milk drink, Group 2 received a formulation with vitamins, minerals, protein and fibre that they added to a control milk and Group 3 received 10 million CFU of B. coagulans Unique IS-2 alongside the vitamin/mineral/protein and fibre formulation which they added to the control milk. Participants in each group took their respective milk drink daily for eight weeks. Natural killer (NK) cell activity was assessed. NK cells are white blood cells which play a critical role in defence, especially against viral infections.
At the end of the study, NK cell activity improved significantly in Group 3 (with B. coagulans Unique IS-2) compared to Group 1 and Group 2. NK activity had nearly doubled in Group 3; in men, activity had increased from 7.1 to 13.3 and in women activity increased from 6.1 to 12.6. After further analysis, it was found that 56% of individuals in Group 3 experienced over 200% improvement in NK cell activity compared with 24% in Group 2 and 5% in Group 1. These results show promise for B. coagulans Unique IS-2 to enhance immune response (Upadhyaya and Banerjee, 2011).
Researchers are just starting to investigate the fascinating and complex pathways between the gut and liver (gut-liver axis). It is hypothesised that improving tight junctions in the gut and reducing bacterial translocation and overgrowth, ethanol production and inflammation may play a vital role in liver health. There is a strong association between cirrhosis and bacterial translocation (Gratz, Mykkanen and El-Nezami, 2010). This suggests that probiotic intervention may offer promise to those with liver disease including cirrhosis.
A gold standard study investigated the use of B. coagulans Unique IS-2 in 65 patients with liver cirrhosis over a six month period. Participants were divided into two groups; Group 1 received a placebo and Group 2 received 2 billion CFU of B. coagulans Unique IS-2 twice daily. In the probiotic group, there were significant reductions in plasma ammonia, hepatic encephalopathy and hospital admissions. Serum bilirubin levels did worsen in both groups; however, this was significantly less in the probiotic group. Overall, results highlight the potential of B. coagulans Unique IS-2 to reduce the risk of major complications associated with cirrhosis (Pawar R, Pardeshi and Ghongane, 2012).
Bacterial vaginosis (BV) is an intimate health condition associated with vaginal dysbiosis. Typically, reduced levels of lactobacilli and higher numbers of potential pathogens such as Gardnerella spp. or Escherichia coli are observed. It can cause symptoms such as itching, burning and odours and affect quality of life. Specific probiotics have been extensively researched for BV and shown positive results. Due to the anti-microbial effects of B. coagulans Unique IS-2, a group of scientists investigated its use alongside conventional treatment for BV.
A controlled study was conducted in 40 women with diagnosed BV. Participants were divided into two groups; a control and probiotic. The control group were given standard antibiotic treatment (Ofloxacin–Ornidazole) for five days alongside a vaginal pessary (co-kimaxazol) for three days. The probiotic group received both treatments and also took 2 billion CFU of B. coagulans Unique IS-2 twice daily for 90 days (orally).
Odour and discharge significantly improved in the probiotic group compared to the control group. Approximately 80% of individuals in the probiotic group were negative for the amine (Whiff) tests compared with 45% in the controlgroup. This was the same for discharge where 80% of women in the probiotic group reported no unusual discharge by 90 days. Burning, itching and inflammation (redness) also improved in the probiotic group. Overall, this preliminary study highlights potential for B. coagulans Unique IS-2 in intimate health but morestudies are necessary for further evaluation (Ratna Sudha, Yelikar and Deshpande, 2012).
Intense exercise induces a degree of gastrointestinal (GI) damage and as a result can impair protein and carbohydrate absorption. Studies have demonstrated the beneficial effects of probiotics in improving the digestibility and absorption of proteins.
A double-blind RCT involving 70 healthy resistance-trained male adults, with a mean age of 21 years, investigated the effect of probiotic supplementation on protein absorption. Participants were divided into two groups for the duration of the 60-day study - a probiotic group and a placebo group. The probiotic group were given 2 billion CFU of Bacillus coagulans Unique IS-2 + 20g Whey protein concentrate (15.4g protein) once a day. The placebo group were given Whey protein concentrate + lactose.
Each participant followed a controlled diet (50% carb: 25% protein: 25% fat) with 1.4g protein per kg body weight + 15.4g protein from Whey protein concentrate per day. All participants also followed the same resistance training protocol during the study period, including full body workouts 4 times per week.
Results from the study found that a significant improvement in muscle strength and muscle power was observed in the Bacillus coagulans Unique IS-2 probiotic group compared to the placebo group. This was shown by a 16.6% increase in one Repetition Maximum (RM) of leg press (p = 0.024) and 7.86% increase in vertical jump power (p = 0.007), respectively.
Additionally, total plasma free amino acid levels were significantly increased by 16.1% (95% CI: 5.1-27.1%, p= 0.005) in the probiotic group compared to placebo group. (Tariq et al, 2022)
Author: Information on this strain was gathered by Dr Kate Steed PhD Food and Microbial Sciences; Gut Microbiology (University of Reading), BSc Medical Microbiology.
For more information and the latest research on probiotics, please visit the Probiotic Professionals pages.
For products containing this strain please visit the Optibac shop.
Ahire, J. J., Neelamraju, J. and Madempudi, R. S. (2020) ‘Behavior of Bacillus coagulans Unique IS2 spores during passage through the simulator of human intestinal microbial ecosystem (SHIME) model’, LWT, 124, p. 109196. doi: 10.1016/j.lwt.2020.109196.
Asgarshirazi, M., Shariat, M. and Dalili, H. (2015) ‘Comparison of the Effects of pH-Dependent Peppermint Oil and Synbiotic Lactol (Bacillus coagulans + Fructooligosaccharides) on Childhood Functional Abdominal Pain: A Randomized Placebo-Controlled Study’, Iranian Red Crescent Medical Journal, 17(4), pp. e23844-undefined. doi: 10.5812/ircmj.17(4)2015.23844.
Azimirad, M., Alebouyeh, M. and Naji, T. (2017) ‘Inhibition of Lipopolysaccharide-Induced Interleukin 8 in Human Adenocarcinoma Cell Line HT-29 by Spore Probiotics: B. coagulans and B. subtilis (natto)’, Probiotics and Antimicrobial Proteins, 9, pp. 56–63.
Gratz, S. W., Mykkanen, H. and El-Nezami, H. (2010) ‘Probiotics and gut health: A special focus on liver diseases’, World Journal of Gastroenterology, 16(4), pp. 403–410. doi: 10.3748/wjg.v16.i4.403.
Khanbhai, A. and Singh Sura, D. (2013) ‘Irritable bowel syndrome for primary care physicians’, British Journal of Medical Practitioners , 6(1), pp. a608-undefined.
Madempudi, R. et al. (2020) ‘Bacillus coagulans Unique IS2 in Constipation: A Double-Blind, Placebo-Controlled Study’, Probiotics and Antimicrobial Proteins , 12, pp. 335–342.
Madempudi, R. S. et al. (2019) ‘Randomized clinical trial: the effect of probiotic Bacillus coagulans Unique IS2 vs. placebo on the symptoms management of irritable bowel syndrome in adults’, Scientific Reports, 9(1). doi: 10.1038/s41598-019-48554-x.
Pawar R, Pardeshi, M. and Ghongane, B. (2012) ‘Study of Effects of Probiotic Lactobacilli in Preventing Major Complications in Patients of Liver Cirrhosis.’, Intl J Biomed Pharma Sci:, 3(1), pp. 206–211.
Ratna Sudha, M., Yelikar, K. A. and Deshpande, S. (2012) ‘Clinical Study of Bacillus coagulans Unique IS-2 (ATCC PTA-11748) in the Treatment of Patients with Bacterial Vaginosis’, Indian Journal of Microbiology, 52(3). doi: 10.1007/s12088-011-0233-z.
Robin, S. G. et al. (2018) ‘Prevalence of Pediatric Functional Gastrointestinal Disorders Utilizing the Rome IV Criteria’, The Journal of Pediatrics, 195, pp. 134–139. doi: 10.1016/j.jpeds.2017.12.012.
Saneian, H. et al. (2013) ‘Comparison of Lactobacillus Sporogenes plus mineral oil and mineral oil alone in the treatment of childhood functional constipation’, J Res Med Sci, 18(2), pp. 85–8.
Saneian, H. et al. (2015) ‘Synbiotic containing Bacillus coagulans and fructo-oligosaccharides for functional abdominal pain in children’, Gastroenterol Hepatol Bed Bench, 8(1), pp. 56–65.
Sudha, M. and Arunasree, K. (2015) ‘ANTI-INFLAMMATORY AND IMMUNOMODULATORY EFFECTS OF BACILLUS COAGULANS UNIQUE IS2’, International Journal of Probiotics & Prebiotics, 10(1), pp. 31–36.
Sudha, M. R. et al. (2018) ‘Efficacy of Bacillus coagulans Unique IS2 in treatment of irritable bowel syndrome in children: a double blind, randomised placebo controlled study’, Beneficial Microbes, 9(4), pp. 563–572. doi: 10.3920/BM2017.0129.
Sudha, R. and Bhonagiri, S. (2012) ‘Efficacy of Bacillus coagulans strain Unique IS-2 in the treatment of patients with acute diarrhea’, Int. J. Probiotics Prebiotics, 7, pp. 33–37.
Tariq, M. et al. (2022) ' The efect of Bacillus coagulans Unique IS‑2 supplementation on plasma amino acid levels and muscle strength in resistance trained males consuming whey protein: a double‑blind, placebo‑controlled study', European Journal of Nutrition, 61(5), pp. 2673-2685. doi:10.1007/s00394-022-02844-9.
Upadhyaya, S. and Banerjee, G. (2011) ‘ENHANCEMENT OF NATURAL KILLER CELL ACTIVITY IN IMMUNO-COMPROMISED ELDERLY SUBJECTS BY BACILLUS COAGULANS’, International Journal of Probiotics and Prebiotics, 6.